Science news - molecular parasitology
27-September-2010
Molecular parasitology
Anyone who names Paul Cézanne as his favourite scientist, has to be a little confused, you might think. The post-impressionist was a master of design, colour and composition. But he wasn’t a scientist. Or was he?
“Maybe not,” says Professor Dave Barry. “But following the impressionists he was taking new knowledge in physics and applying it to painting. Scientists often say that science and art are quite similar. But a scientist is doing it up here."
He raises his hand and touches his head. "While the painter is doing it here and here.”
He touches his head and his heart. “It is digging deeper, I think. As a scientist, I just wish I could learn to harness both those sources in me. It has nothing to do with mysticism.
"It is all about knowledge and the creativity that comes from knowledge. Because of the way scientists are trained, they often tend to think the same way. There are other ways. I have been able to tap into those only rarely, when I was  painting.
painting.
"So for me the great painters, such as Cézanne, were scientists as well as artists.”
Wellcome Centre and orking together
It would seem to follow that collaborations between science and art should be able to produce something new and exciting in science, Professor Barry says. But they almost never do.
“There have been some good projects but little real synergy. I tried it myself once. I wanted to tap into deep scientific principles and phenomena. But I was going too deeply for the artist, while her approaches to painting were beyond me.”
So until the right artist comes along Professor Barry and his group – as well as the Wellcome Trust Centre for Molecular Parasitology, of which he is director – will continue to work in purely scientific ways.
These scientific methods and technologies are growing more sophisticated all the time - and collaboration is still the key. “Here at the Centre we have the biggest concentration of parasitologists in Europe," he says.
"Our aim is to try to understand how parasites work, spread and interact with their hosts. We want to use that knowledge to develop drugs, vaccines and diagnostics.”
Devastation
The malaria parasite and sleeping sickness trypanosomes are the main focus of interest, says Professor Barry. “Trypanosomes are responsible for much of the devastation caused by disease all over sub-Saharan Africa."
Better understanding of how these parasites get around the defence systems of their hosts will help greatly in fighting the diseases, he says. "These are very clever and very complicated. We now know, for instance, that trypanosomes have 2000 genes for making their protective coats.
"It is almost incredible that the genome could have made so many genes and made them all different. The parasites can combine the genes for their coats in an endless variety of ways. And it's mutating those genes all the time.
"The host will develop antibodies that destroy a population of parasites in his body that have one particular coat. But a small number of parasites in that population will have different coats. They then multiply and the host's immune system has to start all over again."
Meanwhile the person - or valuable livestock - is still suffering badly with the disease. So trying to beat this powerful natural system sounds like an almost hopeless task.
Grounds for optimism

Despite the difficulties Professor Barry is hopeful that the war against the disease parasites can be won, he says. "People have made predictions about beating deadly diseases before. And here we are twenty or thirty years on and all we have found is complexity,"
But the new optimism is based firmly on understanding that complexity and the real world, he says."These tropical diseases kill mainly poor people, so there's no money in them for the drugs companies. That's how commerce works. It would cost too much and take too long to develop an effective drug against them."
There is an alternative. "We are beginning to see academic centres developing drugs. We've been approached for instance by an institute in Korea that wants to use our understanding of the biology to discover and make drugs.
"We've also been working with the Dundee Drug Discovery Unit - also funded by the Wellcome Trust - which has come up with a new drug for sleeping sickness.
Tackling complexity
One promising approach is to develop drugs to attack the processes that change the trypanosome parasite’s coats, says Professor Barry. "We're doing a lot of work to find mechanisms associated with the coat genes. We are studying the molecules and learning more about them.
"The biochemistry of that is very different to the host's. So the drug could be specific, which means it could avoid the side-effects that drugs often have when they hit processes that are similar in host cells.”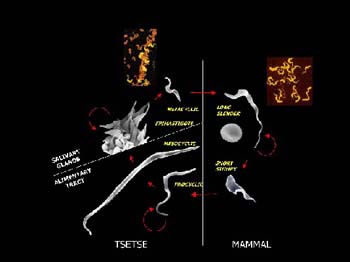
One of the most promising new approaches arises from collaboration with ecologists and mathematicians, Professor Barry believes. Initial doubts about the theoretical basis of these approaches is rapidly giving way to excitement at the possibilities they bring.
“It has been an eye-opener for us looking at transmission, re-infection, how the parasite behaves in the field and interacts with the host. I never thought we would be able to ask some of these large questions, never mind answer them. These are very powerful new methods.
“As one of my ecology colleagues says – it doesn’t matter if it’s a population of elephants on the plains or a population of molecules in a cell. We can model them."
More help with words
| antibodies | breed | cell | chemistry | conception | DNA | fertile |
| fertilisation | fundamental | gene | hosts | immunity | Impressionism | inherit |
| membrane | micro-organism | molecule | observed | parasite | process | protein |
| protoplasm | research | sequence | single-celled | species | structure | tropics |
For other websites and resources relevant to this science story try the

