Science news - connective tissue and dumpy worms
25-Septembert-2010
Dumpy worm
The reason we are not all shapeless blobs is connective tissue, says Iain Johnstone. “That’s what gives our bodies their shape and structure. Every organ in our bodies is held together by connective tissue. Without it we would be a soup of cells.
“So we know how connective tissue can hold cells together. But how does it hold one of the chambers of your heart or the complex structures in a kidney in exactly the right shape to do their job? That is the big question.
“Connective tissue is much more than just a glue. It’s the way it glues cells in a coordinated way. The term is morphology. So how do tissues end up with the correct morphology? There is a huge gap in our knowledge there.”
Morphology is hard for the scientists. It is also very complicated for the developing organism.
No mirror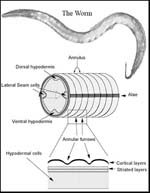
The building blocks of connective tissue are huge molecules secreted by cells. Which then somehow assemble themselves outside the cells. “An engineer building a bridge will work out how to put the building blocks together," says Dr Johnstone. "He will step back and look at the structure taking shape.
"But these things have to assemble themselves without conscious input from a mind. The molecular machinery can’t hold itself up to a mirror and check that it’s got the shape correct. It just does it. Shape generation is incredible.”
But the molecular machinery doesn't always get it right. And when things go wrong with connective tissue the results can be disastrous for a body. Our bones for instance have huge amounts of connective tissue. Many of the more severe connective tissue diseases are bone defects, says Dr Johnstone.
"The most crippling is osteogenesis imperfecta – brittle bone disease – in which people have multiple fractures, reduced stature and twisted skeletons.”
Collagens
Much of the research in Dr Johnstone’s group is on connective tissue proteins called collagens. They study these in a little worm called Caenorhabditis elegans – C. elegans for short. "This is about one millimetre long," he says.
"Most animals have many of the same key molecular mechanisms, and the worm has a lifecycle of two and a half days. So you can do a lot of work very quickly. It's a simple model system for studying the processes.”
Understanding a complex system like the manufacture and assembly of collagen is a bit like understanding a motor car, says Dr Johnstone. “You need to know what the separate parts do before you can understand the whole system.”
The component parts of a motor car are pistons, valves, camshafts and so on. In a biological system they are genes and the proteins they encode. The connective tissue assembled from these proteins corresponds to the car – except that its structure is more complex than any car, or indeed any machine built by humans.
Making dumpy
A molecular biologist like Dr Johnstone uses various techniques for studying the components - the pistons, valves and camshafts his system is built from.
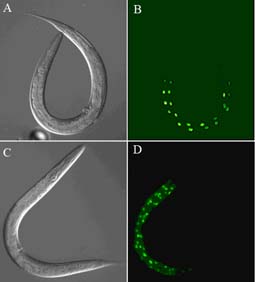
Knocking out a particular gene and seeing what happens to the worm is one of the most common, he says. “Another is immuno -fluorescence detection, which lets us see the shape of a structure.”
Using these methods and others, Dr Johnstone’s team have discovered the precise effect of a particular gene called DPY-7 – pronounced “dumpy 7” – on the little worm C. elegans.
If this gene is knocked out then bands of tissue, like corsets around the worm’s body, fail to develop. These would normally hold it in the correct shape, which should be long and slender.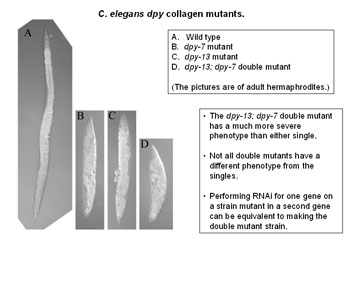
Without this gene the worm can't produce one of the proteins it needs to make its corset. So the corset doesn't get made and the worm grows short, fat and in a word dumpy.
Brittle bones
Dr Johnstone and his colleagues can make dumpy worms in the lab by knocking out the gene. So they know which gene is responsible. But dumpy worms also happen in nature, by mutation. And this is where the research starts to have possible application to humans.
The same type of mutation that makes the worm dumpy causes diseases such as osteogenesis imperfecta - brittle bone disease - in humans. But there is one big difference.
This difference fascinates Dr Johnstone. It offers the possibility of a cure, he believes, for these terrible diseases.
“In humans the mutations are dominant," he explains. "But in C. elegans they are recessive. The defect is the same. But the worm has some capacity to allow the wild type copies of the gene to go out and do their job. We humans lack that ability.
"Now that is interesting. Very interesting indeed. I can’t prove yet that understanding how the worm does it will lead to a cure in humans. But it is certainly worth a shot.”
More help with words
| amino acids | antibodies | cell | complex | conception | DNA | expressed |
| fertilisation | fluorescent | gene | genome | inherit | membrane | organism |
| protoplasm | radiation | sequence | structure | tissue | virus | ultraviolet |
For other websites and resources relevant to this science story try the