Toxoplasma - the perfect parasite
24-Nov-2011
 The perfect parasite
The perfect parasite
There are three people in Markus Meissner's office in the Wellcome Trust Centre for Molecular Parasitology at Glasgow University. So chances are one of us has Toxoplasma gondii living in our body, he says.
At first this sounds like bad news. Toxoplasma is a micro-organism that breeds inside cats. But it doesn't stay there. "It's a parasite with a complicated lifecycle," says Dr Meissner.
"Toxoplasma produces millions of eggs in the cat's intestine, which are then carried out in its faeces. So the eggs are sticking to everywhere the cat does its business."
These eggs find a new home in any warm-blooded animal that swallows them. The usual way into a human body is then through eating the undercooked meat of an animal, such as a sheep or cow, that has swallowed the eggs.
"The eggs hatch in your tummy and the parasite can infect any cell in your body," says Dr Meissner. "Your immune system will kill most of them, but then the parasite can hide in your brain. So you have it for the rest of your life."
Roughly one in three people have Toxoplasma in their bodies, he says. “I was tested when I started working with it. I've got it."
Research
Toxoplasma gondii is not a problem for Dr Meissner or most other people who have it and don't even know. But it can be a serious problem for two  groups - pregnant women or people with immune systems that don’t work well, such as those with AIDS.
groups - pregnant women or people with immune systems that don’t work well, such as those with AIDS.
“If a woman is infected with the parasite for the first time in her life when she is pregnant, it can reach the baby and kill it,” says Dr Meissner. “Toxoplasma is one of the main causes of miscarriage in humans and animals."
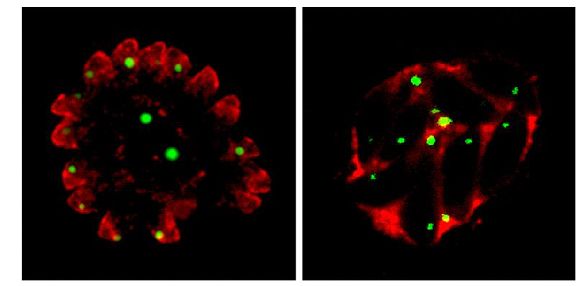
The stage in its complicated lifecycle when Toxoplasma separates into males and females - which then make eggs - happens only in cats. But it can live in the cells of just about every warm-blooded species of animal, says Dr Meissner. "It is very successful. It doesn't usually harm the host.
"It is the perfect parasite, I think."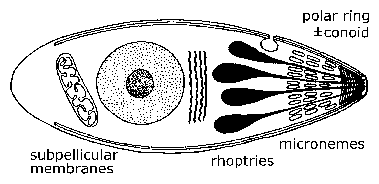
So Dr Meissner's research group at the Wellcome Trust Centre for Molecular Parasitology are investigating the tricks this perfect parasite has learned during evolution. They have a good reason for doing so.
Toxoplasma belongs to a group of parasites called the apicomplexans. It's a large group that includes several much nastier than Toxoplasma - including plasmodium, which causes malaria.
Here's why
Toxoplasma gondii is easier to work with than other members of the group to which it belongs - the apicomplexans, says Dr Meissner. "It is very easy to culture in the lab and to do genetic modifications. It is a close relative of plasmodium. So we can learn about one by studying the other."
Plasmodium is a very small cell because it has to live inside red blood cells, he says. "Toxoplasma is much bigger so we can see more detail in our microscopes. We are using Toxoplasma to try to answer fundamental biological questions about the apicomplexa. Why they can do what they do. How they invade the host cell. 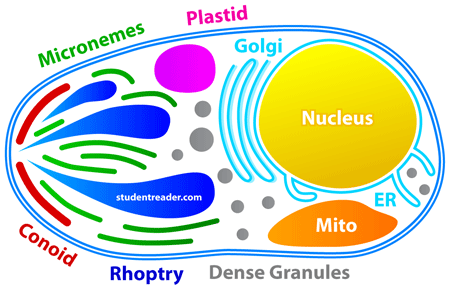
"When the apicomplexans move into a host cell they use its resources to replicate themselves. They have unique structures and organelles that enable them to do that."
Learning how these structures work and how they evolved is one of the main aims of Dr Meissner's research, he says. "If we can really understand the biology we might be able to find an Achilles' Heel. That could then be used not just against Toxoplasma but against other apicomplexans, especially plasmodium."
Here's how
Apicomplexans such as Toxoplasma gondii have special mechanisms inside them that enable them to move around and to invade other cells. Understanding these is the first aim of his research, says Dr Meissner.
The next step is to think up and test chemical methods of interfering with the mechanisms. And because the mechanisms don't exist in the host cell, the chemicals should not interfere with those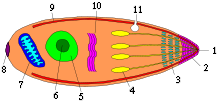 . "A drug that only affects the parasite and not the host cell would be a very good medication," says Dr Meissner.
. "A drug that only affects the parasite and not the host cell would be a very good medication," says Dr Meissner.
So how does Dr Meissner's group explore the nuts and bolts of Toxoplasma's machinery? A common technique in molecular biology is to remove a gene - to "knock it out" - and study what happens to the organism.
But this won't work with Toxoplasma. "Unlike most cells in most animals the parasite only has one copy of each gene," explains Dr Meissner. "The scientific word for that is 'haploid'. So if you remove an essential gene the parasite simply dies before it can do anything. And you don't know why."
So while working for his PhD at Heidelberg University, Dr Meissner (who wasn't a "Dr" then) developed a kind of switch for Toxoplasma genes. "That's what kickstarted my scientific career," he says.
Kickstart switch
So what's the difference between a switch for a gene and a knock-out of a gene? Well a switch lets you decide when, in the parasite's complicated lifecycle, you want to stop the gene working. You can even switch it back on again, says Dr Meissner.
"So I can switch a gene off when the parasite is at the intracellular stage. If it survives I know the gene has no function during that stage. Then I could try switching it off when the parasites are outside the host cell. If I then find they can't invade the cell, I know that gene is needed for invasion.
"It's like taking one part after another out of a car engine while it's still running. You learn what the different parts of the machine do. A cell is a kind of machine. But it's much more complicated than any machine made by humans."
That's what appealed in fact to the young Markus when he first decided to study biology, he says now. "I could always imagine how a car works. If my father bought me a remote-controlled car I'd have one hour of fun playing with it, two hours to disassemble it and never get it back together again. It used to make him crazy.
"I've always loved trying to understand how things work. But how a snail or a Toxoplasma parasite can glide is much more mysterious than how a car can drive."
Words used in pop-ups
| absorb | breed | cell | chemistry | chromosome | degree | diploid |
| DNA | evolution | experiment | expulsion | fertile | foetus | HIV |
| host | inherit | membrane | molecule | process | protein | protoplasm |
| reptile | researcher | sperm | structure | temperature | template | womb |
For other websites and resources relevant to this science story try the

