Parasite sex
20-Sep-2011
Parasite sex
Plasmodium has much more sex than anybody knew, says Dr Lisa Ranford-Cartwright. "They do it all the time.
 "Not all of it is meaningful sex, though. They mate with themselves a lot."
"Not all of it is meaningful sex, though. They mate with themselves a lot."
She demonstrates with a couple of fluffy green models of plasmodium - the parasite that causes malaria - that she happens to have on the desk in her office at Glasgow University's Wellcome Trust Centre for Molecular Parasitology.
"These are amazing, aren't they?" she says. "There's this company that makes all kinds of microbes that look cute and furry - which of course they're not. My lab bought these for me."
The model parasites should really be of different colours, she says, to show better what she's trying to explain. "Then I could say the two green ones can have sex together or the green one can have sex with the blue one."
She thinks for a moment then chuckles. "Maybe I should put lipstick on this one."
Lifecycle
The whole point of sex is to mix one individual's genes 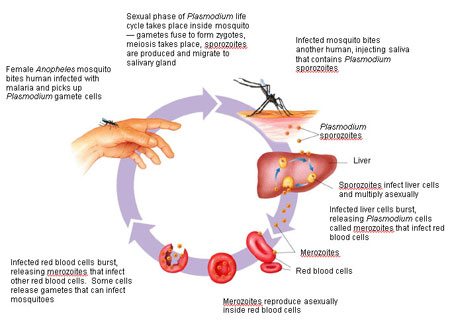 with another's. So it seems pointless for plasmodium to be having sex with itself. The reason, Lisa explains, lies in the complicated lifecycle of the malaria parasite.
with another's. So it seems pointless for plasmodium to be having sex with itself. The reason, Lisa explains, lies in the complicated lifecycle of the malaria parasite.
"To get from one human to another they have to go through a mosquito," she says. "They get into that mosquito when it takes a blood meal from an infected person or animal. Then they have sex in its gut. They mate."
The new plasmodium organisms this creates travel to the next stop on their journey - the mosquito's salivary glands. They don't stay there long. Because a mosquito doesn't just bite and suck blood.
She also injects her saliva into the blood after a bite. This contains an anticoagulant that keeps the blood flowing. (It is always a she, by the way - male mosquitoes eat nectar, not blood.)
Once in the new host's bloodstream, the plasmodium cells are carried by it to the liver. There they feed, grow and divide many times, before being released back into the blood.
They now invade the red blood cells, where they feed, grow and divide many times again.
In this way a person infected with malaria can end up with millions upon millions of plasmodium cells in their blood. These have all come from just a few dozen cells that got into their body through the mosquito bite.
Meaningful sex
If these few dozen plasmodium cells from the mosquito have exactly the same genes as each other, then so too will the new cells in the liver and blood. Like millions upon millions of identical twins.
plasmodium cells from the mosquito have exactly the same genes as each other, then so too will the new cells in the liver and blood. Like millions upon millions of identical twins.
When Lisa began her PhD at Edinburgh University, most researchers thought that's what happened. "They believed people were usually infected with just one parasite type.
"But my research and other people's has begun to unveil the enormous complexity of parasites in people.
"It turns out that in countries where malaria is endemic - such as the whole middle part of Africa - people who are infected usually have at least two unrelated malaria parasites in their bodies.
"In the mosquito's gut those different types can mate with one another or each of them can mate with itself," Lisa says.
"They have a choice. There is some evidence in other organisms that they choose to mate with something different from themselves - to help replace faulty genes and for all the other benefits of sex.
"But my research showed that the malaria parasite can't make that choice. It seems to be random whether it mates with itself or with another."
Good mates
The fact that plasmodium can't choose to mate with another plasmodium that has different genes to itself is a small piece of good news, Lisa says. 
"If they could you'd get even more parasites with new diversity. That would make it much harder for your immune system.
"But the fact is they have a lot of sex and create a lot of diversity even without actively seeking different types of mate. New parasites with new combinations of genes are being made all the time."
Lisa and her group study this diversity. "We want to know what differences in the genotype produce important differences in the phenotype. How do differences in the DNA cause differences in behaviour, appearance, morphology or some other aspect of the parasite?"
They do this research by mating plasmodia and studying the results - "just as farmers mate their cows," Lisa says. "If we have one parasite that multiplies fast, for instance, and another that multiplies slowly, we mate them together and study the offspring.
"Some of these grow fast; some more slowly; some are intermediate. We then look in the genome of these parasites to see which bits are the same in all those that grow at the same rate. In that way we can home in on the genes for fast growth - or any other phenotype we're interested in."
In the field
A plasmodium that multiplies fast in your body makes you much sicker than one that multiplies slowly, Lisa explains. "If I caught malaria I might get a relatively mild infection - it would be nasty but I wouldn't die. Or I might get severely ill and I could die.
"I'm the same person. My immune system is the same. The difference is in the plasmodium."
Other differences in the plasmodium that make malaria more severe - besides multiplying fast - include the ability to stick in the brain and to infect mosquitoes easily.
Lisa's research is aimed at finding all the plasmodium genes that in some way make malaria infections more severe.
"If 100 children come into a clinic with malaria, we know that 98 will recover," she says. "But two will get very sick and will have a high chance of dying. Right now we can't tell which two."
Finding the plasmodium genes that make malaria severe will allow a quick test on a drop of blood from a child to tell how bad the illness is likely to be. Medical help can then concentrate on those who are in most danger.
Around 250 million people catch malaria every year. Nearly a million of those die. Most are young children. Close to 2000 children aged five and under die of malaria in Africa, every day.
"I first went out there in 1990," Lisa says. "You think you know what poverty is before you go. But you don't. You talk to families and they tell you it's normal to have one child, and sometimes two or three, who have died of malaria..
"You remember the long queues of sick people outside the clinics. You remember the children. It changes the way you think about the disease.
"What we do is fascinating science. But it's about more than that. It's about saving lives."
Words used in pop-ups
| absorb | ancestor | bacteria | breed | cell | characteristic | degree |
| descended from | digest | digestive | DNA | embryo | fertilisation | environment |
| experiments | expression | fertile | gene | gland | host | inherit |
| intestine | lubricate | membrane | molecule | organ | pregnancy | process |
| protein | protoplasm | reproductive | species | structure | template | virus |
| womb | zygote |
For other websites and resources relevant to this science story try the